|
VISUAL PHYSICS ONLINE 7.1 THE NATURE OF LIGHT
ELECTROMAGNEITC WAVES P71 004 (A) The
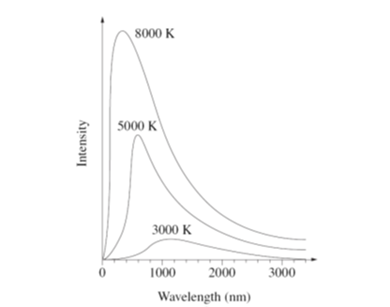
family of curves below shows the relationship between the intensity of
blackbody radiation and its wavelength for various Kelvin temperatures. Who
was the first to correctly explain this relationship? What information do the
graphs give you?
(B) Scientists tried to explain
observations of blackbody radiation using classical wave theory and then
quantum theory. (B1) What is meant by the term blackbody radiation? (B2)
What was a limitation of
classical wave theory that could not explain blackbody radiation? (B3) How does quantum theory satisfactorily explain
blackbody radiation? View solution below only after you have completed the answering the question. |
|
Solution (A) Max Planck was the first person to give a qualitative explanation of the curves for the radiation emitted by a blackbody. He assumed that energy is quantized. The change in energy of an oscillator is a nhf where n is an integer. The total area under the curves represents to energy emitted by the blackbody. The higher the temperature, the more energy radiated. Wien’s Displacement Law lpeak T = constant The higher the temperature, the shorter the wavelength of the em radiation emitted by the blackbody. (B1) Blackbody - object which gives the maximum amount of energy radiated from its surface at any temperature and wavelength and the absorbs all the radiation that falls on it. (B2) JWS Rayleigh
& James Jeans Math model described blackbody radiation curves at long wavelengths accurately but not at shorter wavelengths in the UV, radiation from cavity absorbed and emitted with ever smaller and smaller l, energy emitted from cavity becoming infinite – UV catastrophe (B3) Max Planck was the first person to give a qualitative explanation of the curves for the radiation emitted by a blackbody. He assumed that energy is quantized. The change in energy of an oscillator is a nhf where n is an integer. |